Post by tomsylver on Dec 13, 2017 9:47:10 GMT
Tesaro: Updates To Thesis
Dec. 7, 2017
Jonathan Faison
Summary
* Shares have lost a quarter of their value since my latest update piece, and technicals continue to look weak.
* Concerns with competition appear to be the main factor weighing on the stock.
* Clinical updates, including with its PD-1 molecule and niraparib expansion studies, could also drive upside in 2018.
* I look favorably on the company's ability to obtain much-needed non-dilutive financing.
* Regulatory catalysts and sales growth could also result in shares bouncing back.
Shares of Tesaro (TSRO) have lost a quarter of their value since my latest update piece on August 11th where I stated that strong Zejula sales bode well but concerns (namely competition) remained.
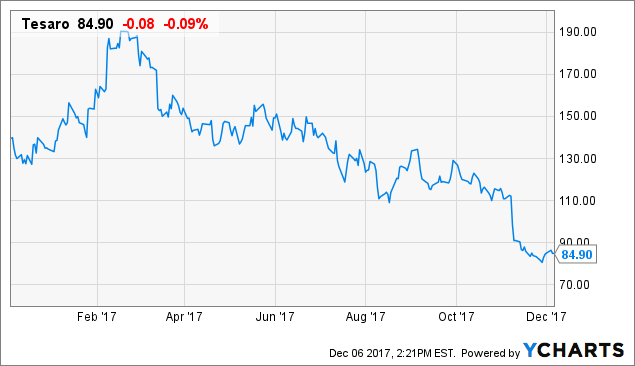
Chart TSRO data by YCharts
With shares at lows going into the end of the year, I believe it's important to review the bullish thesis and recent developments to see where the stock stands now.
In the update piece, I was encouraged by growing Zejula sales along with its 60% slice of the market, making it the most prescribed PARP inhibitor in the United States (an eyebrow-raising claim made by management). I was hopeful for EU approval in the near term with a launch to follow. Additionally, I looked ahead to expansion studies into new indications. These included:
*PRIMA trial in first-line ovarian cancer.
*QUADRA trial for the treatment of patients with ovarian cancer who have received three or more prior lines of chemotherapy.
*TOPACIO trial combined with Keytruda in patients with platinum resistant ovarian cancer or with triple negative breast cancer.
*AVANOVA trial combined with bevacizumab in patients with recurrent ovarian cancer.
I also believed the company's immuno-oncology assets to be completely overlooked, as TSR-022 (anti-TIM-3) and TSR-033 (anti-LAG-3) continued to progress in early-stage studies. Lastly I noted that the oral formulation of Varubi was the most prescribed oral NK-1 receptor antagonist in the United States during the quarter and looked forward to its October PDUFA date.
In mid-August shares were under more pressure after AstraZeneca's (AZN) Lynparza received a broader label from the FDA. The drug can now be used for the maintenance treatment of adult patients with recurrent epithelial ovarian, fallopian tube or primary peritoneal cancer, who are in a complete or partial response to platinum-based chemotherapy.
In early September, we got a look at early data for its PD-1 molecule TSR-042 presented at the annual ESMO meeting. In the first part of the phase 1 study, 2 of 21 patients with ovarian and small cell lung cancer treated with the drug candidate experienced a partial response and five patients with fallopian or ovarian cancer had stable disease. I look forward to future data for further clarification on the drug candidate's prospects.
Also in mid-September, the company received a positive opinion from the CHMP (Committee for Medicinal Products for Human Use) for Zejula as a monotherapy for the maintenance treatment of adult patients with platinum-sensitive relapsed high grade serous epithelial ovarian, fallopian tube, or primary peritoneal cancer who are in a complete response or partial response to platinum-based chemotherapy. On November 20th European approval came as expected. As noted before, Zejula is the first entrant into the European market to be approved for use in patients without regard to BRCA mutation or biomarker.
In late October, Varubi IV crossed the finish line when it received approval from the FDA in combination with other antiemetic agents in adults for the prevention of delayed nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy, including but not limited to highly emetogenic chemotherapy. However, I only expect peak sales in the range of $100 million to $150 million (conservative measure). For the sake of reference, Merck's (NYSE:MRK) Emend had sales of $276 million for the first half of 2017.
In November, the company secured much-needed access to $500 million of non-dilutive term loan financing from investment funds managed by Pharmakon Advisors. The way it will work is that $300 million can be accessed in early December, followed by up to $200 million if needed until December 20th next year.
For the third quarter, the company reported cash and equivalents of $521.3 million, while net loss for the quarter came in at $25.3 million. Research and development expenses rose to $73.4 million, and selling, general and administrative expenses more than doubled to $84.0 million. License, collaboration and other revenue amounted to $101 million with almost all of it due to the up-front payment received from Takeda (OTCPK:TKPYY). Zejula revenues totaled $39.4 million and Varubi revenues came in at $2.4 million.
As for material near-term catalysts, there are several. Continued growth in Zejula sales is expected with management projecting full-year net revenue between $105 million and $115 million. I note that peak sales estimates are for around $2 billion. Management has guided for Zejula to be launched in the UK and Germany in December with additional European countries to follow in the near term.
As for the pipeline in 2018, we can expect data from the TOPACIO study, initial data for the AMBER study evaluating TSR-022 in combination with TSR-042 and initiation of a few mid-and-late stage studies of niraparib in first-line ovarian cancer (in combination with bevacizumab and in combination with TSR-042) and in NSCLC (combination with TSR-042).
Tesaro is a Buy.
Readers who have done their due diligence and are interested in the story should purchase a pilot position in the near term. I would look for technical strength and continued positive developments to add to one's stake. I would be wary of continued weakness which could signal that the sellers still have the upper hand in this one. More conservative readers might want to stay on the sidelines until the competitive dynamics of this market are more readily understood.
There are several risks to the investing thesis - thanks to its recent non-dilutive financing agreement, dilution does not appear likely in the near term. Disappointing results or setbacks with expansion studies of Zejula as well as other immuno-oncology assets or its PD-1 molecule would weigh on shares. Wall Street wants to see growth continue and accelerate for Zejula, so any slowdown in the launch would be high disappointing as well.
As mentioned before, competition from the likes of AstraZeneca and Clovis Oncology (CLVS) is a chief concern and it remains to be seen how this dynamic plays out.

Figure 2: Competitor Lynparza growth and catalysts (Source: AstraZeneca Q3 presentation)
Author's note: My goal is to bring to readers' attention undervalued stocks with catalysts that could propel shares higher, as well as provide a fresh perspective on stocks you may already be aware of. I also touch on planning trades and risk management, as those are two areas I feel are often neglected. If you found value in the above article, consider clicking the orange "Follow" button and getting email alerts to receive my latest content. My sincere appreciation for readers who add value and join the discussion in the comments section, as well as those who share my work with others who could benefit from it.
Disclaimer: Commentary presented is not individualized investment advice. Opinions offered here are not personalized recommendations. Readers are expected to do their own due diligence or consult an investment professional if needed prior to making trades. Strategies discussed should not be mistaken for recommendations, and past performance may not be indicative of future results. Although I do my best to present factual research, I do not in any way guarantee the accuracy of the information I post. Investing in common stock can result in partial or total loss of capital. In other words, readers are expected to (and encouraged) form their own trading plan, do their own research and take responsibility for their own actions. If they are not able or willing to do so, better to buy index funds or find a thoroughly vetted fee-only financial advisor to handle your account. I am in a collaborative relationship with The Biotech Forum/Bret Jensen.
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Dec. 7, 2017
Jonathan Faison
Summary
* Shares have lost a quarter of their value since my latest update piece, and technicals continue to look weak.
* Concerns with competition appear to be the main factor weighing on the stock.
* Clinical updates, including with its PD-1 molecule and niraparib expansion studies, could also drive upside in 2018.
* I look favorably on the company's ability to obtain much-needed non-dilutive financing.
* Regulatory catalysts and sales growth could also result in shares bouncing back.
Shares of Tesaro (TSRO) have lost a quarter of their value since my latest update piece on August 11th where I stated that strong Zejula sales bode well but concerns (namely competition) remained.

Chart TSRO data by YCharts
With shares at lows going into the end of the year, I believe it's important to review the bullish thesis and recent developments to see where the stock stands now.
In the update piece, I was encouraged by growing Zejula sales along with its 60% slice of the market, making it the most prescribed PARP inhibitor in the United States (an eyebrow-raising claim made by management). I was hopeful for EU approval in the near term with a launch to follow. Additionally, I looked ahead to expansion studies into new indications. These included:
*PRIMA trial in first-line ovarian cancer.
*QUADRA trial for the treatment of patients with ovarian cancer who have received three or more prior lines of chemotherapy.
*TOPACIO trial combined with Keytruda in patients with platinum resistant ovarian cancer or with triple negative breast cancer.
*AVANOVA trial combined with bevacizumab in patients with recurrent ovarian cancer.
I also believed the company's immuno-oncology assets to be completely overlooked, as TSR-022 (anti-TIM-3) and TSR-033 (anti-LAG-3) continued to progress in early-stage studies. Lastly I noted that the oral formulation of Varubi was the most prescribed oral NK-1 receptor antagonist in the United States during the quarter and looked forward to its October PDUFA date.
In mid-August shares were under more pressure after AstraZeneca's (AZN) Lynparza received a broader label from the FDA. The drug can now be used for the maintenance treatment of adult patients with recurrent epithelial ovarian, fallopian tube or primary peritoneal cancer, who are in a complete or partial response to platinum-based chemotherapy.
In early September, we got a look at early data for its PD-1 molecule TSR-042 presented at the annual ESMO meeting. In the first part of the phase 1 study, 2 of 21 patients with ovarian and small cell lung cancer treated with the drug candidate experienced a partial response and five patients with fallopian or ovarian cancer had stable disease. I look forward to future data for further clarification on the drug candidate's prospects.
Also in mid-September, the company received a positive opinion from the CHMP (Committee for Medicinal Products for Human Use) for Zejula as a monotherapy for the maintenance treatment of adult patients with platinum-sensitive relapsed high grade serous epithelial ovarian, fallopian tube, or primary peritoneal cancer who are in a complete response or partial response to platinum-based chemotherapy. On November 20th European approval came as expected. As noted before, Zejula is the first entrant into the European market to be approved for use in patients without regard to BRCA mutation or biomarker.
In late October, Varubi IV crossed the finish line when it received approval from the FDA in combination with other antiemetic agents in adults for the prevention of delayed nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy, including but not limited to highly emetogenic chemotherapy. However, I only expect peak sales in the range of $100 million to $150 million (conservative measure). For the sake of reference, Merck's (NYSE:MRK) Emend had sales of $276 million for the first half of 2017.
In November, the company secured much-needed access to $500 million of non-dilutive term loan financing from investment funds managed by Pharmakon Advisors. The way it will work is that $300 million can be accessed in early December, followed by up to $200 million if needed until December 20th next year.
For the third quarter, the company reported cash and equivalents of $521.3 million, while net loss for the quarter came in at $25.3 million. Research and development expenses rose to $73.4 million, and selling, general and administrative expenses more than doubled to $84.0 million. License, collaboration and other revenue amounted to $101 million with almost all of it due to the up-front payment received from Takeda (OTCPK:TKPYY). Zejula revenues totaled $39.4 million and Varubi revenues came in at $2.4 million.
As for material near-term catalysts, there are several. Continued growth in Zejula sales is expected with management projecting full-year net revenue between $105 million and $115 million. I note that peak sales estimates are for around $2 billion. Management has guided for Zejula to be launched in the UK and Germany in December with additional European countries to follow in the near term.
As for the pipeline in 2018, we can expect data from the TOPACIO study, initial data for the AMBER study evaluating TSR-022 in combination with TSR-042 and initiation of a few mid-and-late stage studies of niraparib in first-line ovarian cancer (in combination with bevacizumab and in combination with TSR-042) and in NSCLC (combination with TSR-042).
Tesaro is a Buy.
Readers who have done their due diligence and are interested in the story should purchase a pilot position in the near term. I would look for technical strength and continued positive developments to add to one's stake. I would be wary of continued weakness which could signal that the sellers still have the upper hand in this one. More conservative readers might want to stay on the sidelines until the competitive dynamics of this market are more readily understood.
There are several risks to the investing thesis - thanks to its recent non-dilutive financing agreement, dilution does not appear likely in the near term. Disappointing results or setbacks with expansion studies of Zejula as well as other immuno-oncology assets or its PD-1 molecule would weigh on shares. Wall Street wants to see growth continue and accelerate for Zejula, so any slowdown in the launch would be high disappointing as well.
As mentioned before, competition from the likes of AstraZeneca and Clovis Oncology (CLVS) is a chief concern and it remains to be seen how this dynamic plays out.

Figure 2: Competitor Lynparza growth and catalysts (Source: AstraZeneca Q3 presentation)
Author's note: My goal is to bring to readers' attention undervalued stocks with catalysts that could propel shares higher, as well as provide a fresh perspective on stocks you may already be aware of. I also touch on planning trades and risk management, as those are two areas I feel are often neglected. If you found value in the above article, consider clicking the orange "Follow" button and getting email alerts to receive my latest content. My sincere appreciation for readers who add value and join the discussion in the comments section, as well as those who share my work with others who could benefit from it.
Disclaimer: Commentary presented is not individualized investment advice. Opinions offered here are not personalized recommendations. Readers are expected to do their own due diligence or consult an investment professional if needed prior to making trades. Strategies discussed should not be mistaken for recommendations, and past performance may not be indicative of future results. Although I do my best to present factual research, I do not in any way guarantee the accuracy of the information I post. Investing in common stock can result in partial or total loss of capital. In other words, readers are expected to (and encouraged) form their own trading plan, do their own research and take responsibility for their own actions. If they are not able or willing to do so, better to buy index funds or find a thoroughly vetted fee-only financial advisor to handle your account. I am in a collaborative relationship with The Biotech Forum/Bret Jensen.
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.




