Post by icemandios on Mar 16, 2022 16:02:32 GMT
March 16, 2022 09:29 AM EDTUpdated 10:48 AM Pharma
Scoop: Biogen will finally publish its PhIII results for Aduhelm
Zachary Brennan
Senior Editor
Biogen quietly announced at a conference in Spain today that a manuscript of the Phase III results for the company’s controversial Alzheimer’s drug Aduhelm will finally be published, almost a year after the drug won its accelerated approval.
The “Phase 3 manuscript was accepted for publication” in the Journal of Prevention of Alzheimer’s Disease, a spokesperson told Endpoints News, pointing to today’s presentation at the 2022 International Conference on Alzheimer’s and Parkinson’s Diseases.
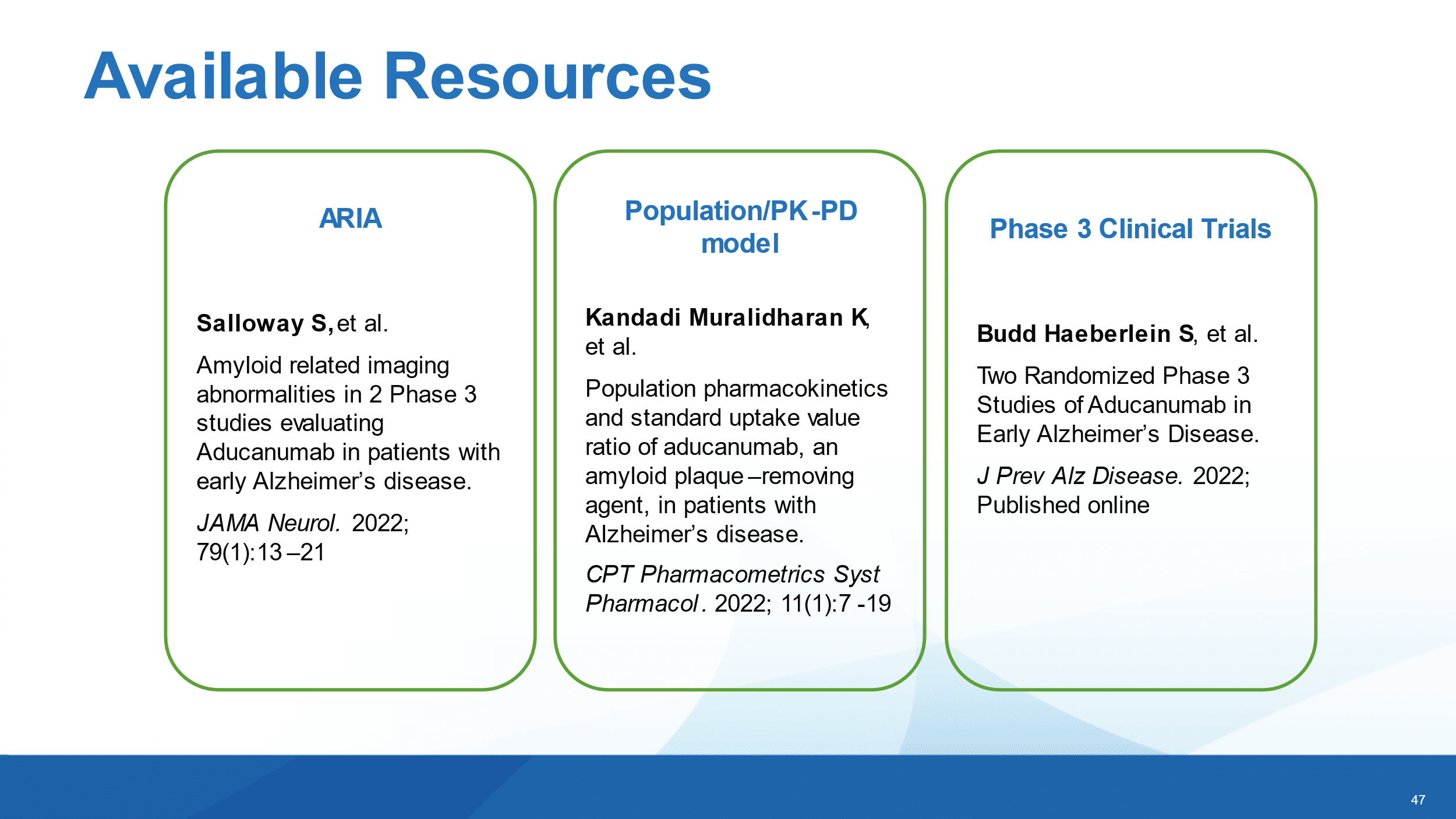
“We are planning to issue a press release as soon as the manuscript is available online,” the spokesperson said.
The data may finally offer researchers and potentially prescribing doctors a better view of how to use Aduhelm and what the safety data show.

Madhav Thambisetty
Madhav Thambisetty, a senior investigator at NIH’s National Institute on Aging, previously told Endpoints that the published data “will enable clinicians like myself in our decision-making. For instance, it would be important to understand whether the drug accelerated brain atrophy in the clinical trial participants. Loss of brain volume has been reported with other amyloid lowering antibodies and while serial MR imaging of the brain was performed in the two phase-3 trials of aducanumab, we do not yet know if this drug results in accelerated brain atrophy in patients.”
While announcing the publication, the company also released new data showing that Aduhelm continues to reduce amyloid beta in the brain for two-and-a-half years (FDA’s accelerated approval was based on 1.5 years of data). But still, the company did not offer additional data on whether this reduction in amyloid can slow cognitive decline.
Scoop: Biogen will finally publish its PhIII results for Aduhelm
Zachary Brennan
Senior Editor
Biogen quietly announced at a conference in Spain today that a manuscript of the Phase III results for the company’s controversial Alzheimer’s drug Aduhelm will finally be published, almost a year after the drug won its accelerated approval.
The “Phase 3 manuscript was accepted for publication” in the Journal of Prevention of Alzheimer’s Disease, a spokesperson told Endpoints News, pointing to today’s presentation at the 2022 International Conference on Alzheimer’s and Parkinson’s Diseases.

“We are planning to issue a press release as soon as the manuscript is available online,” the spokesperson said.
The data may finally offer researchers and potentially prescribing doctors a better view of how to use Aduhelm and what the safety data show.

Madhav Thambisetty
Madhav Thambisetty, a senior investigator at NIH’s National Institute on Aging, previously told Endpoints that the published data “will enable clinicians like myself in our decision-making. For instance, it would be important to understand whether the drug accelerated brain atrophy in the clinical trial participants. Loss of brain volume has been reported with other amyloid lowering antibodies and while serial MR imaging of the brain was performed in the two phase-3 trials of aducanumab, we do not yet know if this drug results in accelerated brain atrophy in patients.”
While announcing the publication, the company also released new data showing that Aduhelm continues to reduce amyloid beta in the brain for two-and-a-half years (FDA’s accelerated approval was based on 1.5 years of data). But still, the company did not offer additional data on whether this reduction in amyloid can slow cognitive decline.




