Leukemia Research Supported by Strategic Investment ZEBRA
Oct 23, 2015 19:27:51 GMT
onefreetrajectory, Chuck-2, and 2 more like this
Post by Uncle on Oct 23, 2015 19:27:51 GMT
The Scripps Research Institute, News & Views
VOL 15. ISSUE 32
October 26, 2015
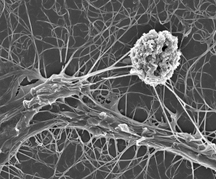
A Natural Killer (NK) cell (left) destroys an acute myeloid leukemia target cell (right). The NK cell, formerly also a myeloid leukemia cell, was induced by four days of treatment with an antibody. (Scanning electron microscopy image courtesy of the Lerner lab.)
Scientists Find Way to Make Leukemia Cells Kill Each Other
Scientists at The Scripps Research Institute (TSRI) have found a way to change leukemia cells into leukemia-killing immune cells. The surprise finding could lead to a powerful new therapy for leukemia and possibly other cancers.
“It’s a totally new approach to cancer, and we’re working to test it in human patients as soon as possible,” said senior investigator Richard A. Lerner, Institute Professor and the Lita Annenberg Hazen Professor of Immunochemistry at TSRI.
The findings, published this week in the Proceedings of the National Academy of Sciences, result from the discovery of a rare human antibody.
Unexpected Effects
The Lerner laboratory has pioneered techniques to generate and screen very large libraries of antibodies (immune system molecules), using the power of large numbers to find therapeutic antibodies that bind to a desired target or activate a desired receptor on cells.
Recently, the lab mounted an effort to find therapies for people with certain immune cell or blood factor deficiencies, by looking for antibodies that activate growth-factor receptors on immature bone marrow cells that might induce these bone marrow cells to mature into specific blood cell types. Over the past few years, Lerner and his team succeeded in identifying a number of antibodies that activate marrow-cell receptors in this way.
In the process, the scientists noted that some of these receptor-activating antibodies have unexpected effects on marrow cells, causing them to mature into radically different cell types, such as neural cells.
While why this happens is an unresolved issue, the discovery led the team to wonder if they could also use the method to convert cancerous marrow cells (leukemia cells) into non-cancerous cells.
Following the Trail
To find out, in the new study Lerner and his team, including first author Kyungmoo Yea, an assistant professor of cellular and molecular biology at TSRI, tested 20 of their recently discovered receptor-activating antibodies against acute myeloid leukemia cells from human patients. One of these antibodies turned out to have an extraordinary impact on the acute myeloid leukemia cells.
A high percentage of acute myeloid leukemia cells express the thrombopoietin (TPO) receptor, and the effective antibody was a highly potent and selective activator of this receptor on marrow cells. When the antibody was applied to healthy immature marrow cells, it caused them to mature into blood-platelet-producing cells called megakaryocytes. However, when the antibody was applied to acute myeloid leukemia cells, they matured into very different cells known as dendritic cells, key support cells in the immune system.
By itself, this could be a valuable therapeutic strategy, but it wasn’t the end of the story. Lerner’s team noted that, with longer exposures to the antibodies and certain other lab-dish conditions, the induced dendritic cells developed further—into cells that closely resembled natural killer (NK) cells.
NK cells represent one of the rapid-reaction forces of the immune system. They can be effective against viruses and bacteria—and cancer cells—even without prior exposure. They don’t have highly specific receptors for recognizing individual targets, as T-cells do, but instead are capable of detecting, in a general way, when a nearby cell is infected or cancerous.
“That antibody could have turned those acute myeloid leukemia cells into a lot of other cell types, but somehow we were lucky enough to get NK cells,” Lerner said.
‘Fratricide’
The team examined these induced NK cells with electron microscopy and observed that many of the cells had extended tendrils through the outer membranes of neighboring leukemic cells—their erstwhile brethren. In lab dish tests, a modest number of these NK cells wiped out about 15 percent of the surrounding acute myeloid leukemia cell population in just 24 hours.
Curiously, the induced NK cells’ cancer-killing effect appeared to be purely fratricidal. The researchers found that unrelated breast cancer cells did not die off in large numbers when in the presence of the NK cells.
Why the induced NK cells appear to target only closely related cells isn’t yet clear. In principle, though, there are yet-to-be-discovered antibodies—and even small-molecule compounds—that would turn other cancerous cell types into fratricidal NK cells, by activating other receptors expressed on those cells.
Such fratricidal therapies, which Lerner terms “fratricidins,” would have several potential advantages. First, especially if they are antibodies, they could be clinically useful with little or no further modification. Second, their high specificity for their target receptors, and the resulting NK cells’ specificity for related cancer cells, should reduce the likelihood of adverse side effects, possibly making them much more tolerable than traditional cancer chemotherapies.
Finally, the peculiar dynamics of fratricidin therapy, in which every cancerous cell is potentially convertible to a cancer-killing NK cell, suggests that—if the strategy works—it might not just reduce the targeted cancer-cell population in a patient, but eliminate it altogether.
“We’re in discussions with pharmaceutical companies to take this straight into humans after the appropriate preclinical toxicity studies,” he said.
Other co-authors of the study, “Agonist antibody that induces human malignant cells to kill one another,” were Hongkai Zhang, Jia Xie, Teresa M. Jones, Chih-Wei Lin, Walter Francesconi, Fulvia Berton, Mohammad Fallahi, and Karsten Sauer, all of TSRI during the study.
The research was supported by the JPB Foundation and Zebra Biologics.
OPKO Strategic Investment Zebra Biologics
OPKO’s Scientific Advisory Board Member, Scientific Founder of Zebra and Senior investigator of the study Richard A. Lerner, M.D.
Dr. Lerner has served as a director of OPKO since March 2007. Dr. Lerner served as President of The Scripps Research Institute, a private, non-profit biomedical research organization, from 1986 until 2011 and is currently serving as an institute professor. Dr. Lerner is a member of numerous scientific associations, including the National Academy of Sciences and the Royal Swedish Academy of Sciences. Dr. Lerner serves as director of Sequenom, Inc. (Nasal: SQNM), a life sciences company. He is also on the Board of Directors for Intra-Cellular Therapies, a privately held biotechnology company, and the board of Teva (NYSE:TEVA). He previously served as a director of Kraft Foods, Inc. and Xencor, a privately held biotechnology company, and on the Siemens’ Advisory Board for Molecular Medicine of Siemens AG. As a result of Dr. Lerner’s long tenure as President of a major biomedical research organization, he provides valuable business, scientific, leadership, and management expertise that helps drive strategic direction and expansion at OPKO. His experience and training as a physician and a scientist enables him to bring valuable advice to the OPKO board, including a critical perspective on drug discovery and development and providing a fundamental understanding of a potential pathways contributing to disease.
At December 31, 2014, OPKO holds an investment in Zebra Biologics, Inc.(“Zebra”), a biotechnology company focused on the discovery and development of biosuperior antibody therapeutics and complex drugs.
10-K:
We acquired 840,000 shares of Zebra Series A-2 Preferred Stock and 900,000 shares of Zebra restricted common stock (ownership 28% at December 31, 2014). Zebra is a privately held biotechnology company focused on the discovery and development of biosuperior antibody therapeutics and complex drugs. Dr. Richard Lerner, M.D., a member of our Board of Directors, is a founder of Zebra and, along with Dr. Frost, serves as a member of Zebra’s Board of Directors.
Zebra Biologics
Zebra Biologics Inc. is a start-up biotechnology company focused on a "next-gen" approach to more efficiently and more effectively discovering and developing novel human biologic drugs utilizing a proprietary platform technology
Zebra was conceived to exploit a technology recently developed at the Scripps Research Institute that represents a quantum leap in unlocking the utility and immense diversity of combinatorial antibody and peptide libraries as sources for deriving new drugs for both new and previously hard to drug targets. The technology is applicable to virtually all disease areas.
Zebra Founders
Dr Richard A Lerner *
Prof., Scripps Res. Inst. (TSRI)
- Past President Scripps Res Inst., Member National Academy Science
- Early pioneer in combinatorial antibody library technology
Dr Ronald M Lindsay *
CEO Zebra Biologics Inc.
- 30 years in pharma- biotech Rx & Dx, Novartis, REGN, MLNM ,SQNM
- Scientific co-founder Regeneron Pharma. Inc.
Dr Phillip Frost *
CEO OPKO Inc.
- Founder and CEO of IVAX Inc. (acquired by Teva)
- Dermatologist, Entrepreneur and Philanthropist, Chairman TEVA
Prof Paul Greengard
Rockefeller University, NYC.
- Renowned neuroscientist, Nobel Laureate in Medicine
- Co-Founderof several biotechs
Dr Guang Yang
Director, SIAIS, Shanghai
- Former GSK scientist and business dev exec. (China)
- Founder and Co-Director Shanghai Antibody Institute (SIAIS)
Mr Feng Gao *
- Chinese Industrialist, Energy Sector
- Lifescience interest in China incl. CRO
* Directors and A-2 Round Investors
Welcome to Zebra - A Future Leader in Antibody Therapeutics
We are exploiting a novel, better, faster, cheaper approach to deriving novel agonist and antagonist fully human biologic drugs from antibody and peptide libraries
Combing The Power Of Numbers With Functional Screens for the Derivation of New Biologics Our singular scientific and clinical goal at Zebra is more effective and more efficient utilization of
the unparalleled diversity of chemical space locked in combinatorial libraries of human antibody genes.
We are achieving this goal by exploiting in a new platform the diversity of the 100 billion+ members of an antibody library with a novel proprietary screening technology that allows new biologic drugs - AGONISTS or ANTAGONISTS - to be selected directly for FUNCTION in human cell based assays.
Appreciating the Numbers of Natural Diversity
Just as we all seem to know that no two snow flakes have the same crystal structure, extensive studies have shown that no two Zebra's have an identical stripe pattern. Hence our name Zebra Biologics.
Drug discovery is all about diversity of chemical space and efficient ways to find rare events - high affinity binding of a novel small molecule, peptide or antibody to a drug target. The combined small molecule libraries of all major pharmaceutical companies are in the range of 50-100 million compounds. The repertoire of antibodies in the standard humanized mouse widely used as a cumbersome in vivo tool for selecting novel antibody candidates for drug development is perhaps 100-500 million. A good combinatorial human antibody library boast 100 billion unique sequences and has the advantage of being exploitable in a test tube for drug discovery.
VOL 15. ISSUE 32
October 26, 2015

A Natural Killer (NK) cell (left) destroys an acute myeloid leukemia target cell (right). The NK cell, formerly also a myeloid leukemia cell, was induced by four days of treatment with an antibody. (Scanning electron microscopy image courtesy of the Lerner lab.)
Scientists Find Way to Make Leukemia Cells Kill Each Other
Scientists at The Scripps Research Institute (TSRI) have found a way to change leukemia cells into leukemia-killing immune cells. The surprise finding could lead to a powerful new therapy for leukemia and possibly other cancers.
“It’s a totally new approach to cancer, and we’re working to test it in human patients as soon as possible,” said senior investigator Richard A. Lerner, Institute Professor and the Lita Annenberg Hazen Professor of Immunochemistry at TSRI.
The findings, published this week in the Proceedings of the National Academy of Sciences, result from the discovery of a rare human antibody.
Unexpected Effects
The Lerner laboratory has pioneered techniques to generate and screen very large libraries of antibodies (immune system molecules), using the power of large numbers to find therapeutic antibodies that bind to a desired target or activate a desired receptor on cells.
Recently, the lab mounted an effort to find therapies for people with certain immune cell or blood factor deficiencies, by looking for antibodies that activate growth-factor receptors on immature bone marrow cells that might induce these bone marrow cells to mature into specific blood cell types. Over the past few years, Lerner and his team succeeded in identifying a number of antibodies that activate marrow-cell receptors in this way.
In the process, the scientists noted that some of these receptor-activating antibodies have unexpected effects on marrow cells, causing them to mature into radically different cell types, such as neural cells.
While why this happens is an unresolved issue, the discovery led the team to wonder if they could also use the method to convert cancerous marrow cells (leukemia cells) into non-cancerous cells.
Following the Trail
To find out, in the new study Lerner and his team, including first author Kyungmoo Yea, an assistant professor of cellular and molecular biology at TSRI, tested 20 of their recently discovered receptor-activating antibodies against acute myeloid leukemia cells from human patients. One of these antibodies turned out to have an extraordinary impact on the acute myeloid leukemia cells.
A high percentage of acute myeloid leukemia cells express the thrombopoietin (TPO) receptor, and the effective antibody was a highly potent and selective activator of this receptor on marrow cells. When the antibody was applied to healthy immature marrow cells, it caused them to mature into blood-platelet-producing cells called megakaryocytes. However, when the antibody was applied to acute myeloid leukemia cells, they matured into very different cells known as dendritic cells, key support cells in the immune system.
By itself, this could be a valuable therapeutic strategy, but it wasn’t the end of the story. Lerner’s team noted that, with longer exposures to the antibodies and certain other lab-dish conditions, the induced dendritic cells developed further—into cells that closely resembled natural killer (NK) cells.
NK cells represent one of the rapid-reaction forces of the immune system. They can be effective against viruses and bacteria—and cancer cells—even without prior exposure. They don’t have highly specific receptors for recognizing individual targets, as T-cells do, but instead are capable of detecting, in a general way, when a nearby cell is infected or cancerous.
“That antibody could have turned those acute myeloid leukemia cells into a lot of other cell types, but somehow we were lucky enough to get NK cells,” Lerner said.
‘Fratricide’
The team examined these induced NK cells with electron microscopy and observed that many of the cells had extended tendrils through the outer membranes of neighboring leukemic cells—their erstwhile brethren. In lab dish tests, a modest number of these NK cells wiped out about 15 percent of the surrounding acute myeloid leukemia cell population in just 24 hours.
Curiously, the induced NK cells’ cancer-killing effect appeared to be purely fratricidal. The researchers found that unrelated breast cancer cells did not die off in large numbers when in the presence of the NK cells.
Why the induced NK cells appear to target only closely related cells isn’t yet clear. In principle, though, there are yet-to-be-discovered antibodies—and even small-molecule compounds—that would turn other cancerous cell types into fratricidal NK cells, by activating other receptors expressed on those cells.
Such fratricidal therapies, which Lerner terms “fratricidins,” would have several potential advantages. First, especially if they are antibodies, they could be clinically useful with little or no further modification. Second, their high specificity for their target receptors, and the resulting NK cells’ specificity for related cancer cells, should reduce the likelihood of adverse side effects, possibly making them much more tolerable than traditional cancer chemotherapies.
Finally, the peculiar dynamics of fratricidin therapy, in which every cancerous cell is potentially convertible to a cancer-killing NK cell, suggests that—if the strategy works—it might not just reduce the targeted cancer-cell population in a patient, but eliminate it altogether.
“We’re in discussions with pharmaceutical companies to take this straight into humans after the appropriate preclinical toxicity studies,” he said.
Other co-authors of the study, “Agonist antibody that induces human malignant cells to kill one another,” were Hongkai Zhang, Jia Xie, Teresa M. Jones, Chih-Wei Lin, Walter Francesconi, Fulvia Berton, Mohammad Fallahi, and Karsten Sauer, all of TSRI during the study.
The research was supported by the JPB Foundation and Zebra Biologics.
OPKO Strategic Investment Zebra Biologics
OPKO’s Scientific Advisory Board Member, Scientific Founder of Zebra and Senior investigator of the study Richard A. Lerner, M.D.
Dr. Lerner has served as a director of OPKO since March 2007. Dr. Lerner served as President of The Scripps Research Institute, a private, non-profit biomedical research organization, from 1986 until 2011 and is currently serving as an institute professor. Dr. Lerner is a member of numerous scientific associations, including the National Academy of Sciences and the Royal Swedish Academy of Sciences. Dr. Lerner serves as director of Sequenom, Inc. (Nasal: SQNM), a life sciences company. He is also on the Board of Directors for Intra-Cellular Therapies, a privately held biotechnology company, and the board of Teva (NYSE:TEVA). He previously served as a director of Kraft Foods, Inc. and Xencor, a privately held biotechnology company, and on the Siemens’ Advisory Board for Molecular Medicine of Siemens AG. As a result of Dr. Lerner’s long tenure as President of a major biomedical research organization, he provides valuable business, scientific, leadership, and management expertise that helps drive strategic direction and expansion at OPKO. His experience and training as a physician and a scientist enables him to bring valuable advice to the OPKO board, including a critical perspective on drug discovery and development and providing a fundamental understanding of a potential pathways contributing to disease.
At December 31, 2014, OPKO holds an investment in Zebra Biologics, Inc.(“Zebra”), a biotechnology company focused on the discovery and development of biosuperior antibody therapeutics and complex drugs.
10-K:
We acquired 840,000 shares of Zebra Series A-2 Preferred Stock and 900,000 shares of Zebra restricted common stock (ownership 28% at December 31, 2014). Zebra is a privately held biotechnology company focused on the discovery and development of biosuperior antibody therapeutics and complex drugs. Dr. Richard Lerner, M.D., a member of our Board of Directors, is a founder of Zebra and, along with Dr. Frost, serves as a member of Zebra’s Board of Directors.
Zebra Biologics
Zebra Biologics Inc. is a start-up biotechnology company focused on a "next-gen" approach to more efficiently and more effectively discovering and developing novel human biologic drugs utilizing a proprietary platform technology
Zebra was conceived to exploit a technology recently developed at the Scripps Research Institute that represents a quantum leap in unlocking the utility and immense diversity of combinatorial antibody and peptide libraries as sources for deriving new drugs for both new and previously hard to drug targets. The technology is applicable to virtually all disease areas.
Zebra Founders
Dr Richard A Lerner *
Prof., Scripps Res. Inst. (TSRI)
- Past President Scripps Res Inst., Member National Academy Science
- Early pioneer in combinatorial antibody library technology
Dr Ronald M Lindsay *
CEO Zebra Biologics Inc.
- 30 years in pharma- biotech Rx & Dx, Novartis, REGN, MLNM ,SQNM
- Scientific co-founder Regeneron Pharma. Inc.
Dr Phillip Frost *
CEO OPKO Inc.
- Founder and CEO of IVAX Inc. (acquired by Teva)
- Dermatologist, Entrepreneur and Philanthropist, Chairman TEVA
Prof Paul Greengard
Rockefeller University, NYC.
- Renowned neuroscientist, Nobel Laureate in Medicine
- Co-Founderof several biotechs
Dr Guang Yang
Director, SIAIS, Shanghai
- Former GSK scientist and business dev exec. (China)
- Founder and Co-Director Shanghai Antibody Institute (SIAIS)
Mr Feng Gao *
- Chinese Industrialist, Energy Sector
- Lifescience interest in China incl. CRO
* Directors and A-2 Round Investors
Welcome to Zebra - A Future Leader in Antibody Therapeutics
We are exploiting a novel, better, faster, cheaper approach to deriving novel agonist and antagonist fully human biologic drugs from antibody and peptide libraries
Combing The Power Of Numbers With Functional Screens for the Derivation of New Biologics Our singular scientific and clinical goal at Zebra is more effective and more efficient utilization of
the unparalleled diversity of chemical space locked in combinatorial libraries of human antibody genes.
We are achieving this goal by exploiting in a new platform the diversity of the 100 billion+ members of an antibody library with a novel proprietary screening technology that allows new biologic drugs - AGONISTS or ANTAGONISTS - to be selected directly for FUNCTION in human cell based assays.
Appreciating the Numbers of Natural Diversity
Just as we all seem to know that no two snow flakes have the same crystal structure, extensive studies have shown that no two Zebra's have an identical stripe pattern. Hence our name Zebra Biologics.
Drug discovery is all about diversity of chemical space and efficient ways to find rare events - high affinity binding of a novel small molecule, peptide or antibody to a drug target. The combined small molecule libraries of all major pharmaceutical companies are in the range of 50-100 million compounds. The repertoire of antibodies in the standard humanized mouse widely used as a cumbersome in vivo tool for selecting novel antibody candidates for drug development is perhaps 100-500 million. A good combinatorial human antibody library boast 100 billion unique sequences and has the advantage of being exploitable in a test tube for drug discovery.





