Chinese Firm is Claiming Success
Nov 4, 2019 16:31:57 GMT
sgard434, miamianne67, and 3 more like this
Post by icemandios on Nov 4, 2019 16:31:57 GMT
November 4, 2019 06:52 AM ESTUpdated 09:49 AM
Amber Tong
China
Regulatory
UPDATED: Alzheimer's stunner: Chinese biotech wins OK for a new drug on positive data — and they have some high-profile supporters
Did an obscure Chinese biotech just crack the Alzheimer’s enigma that’s dumbfounded a host of major league drug developers?
China’s National Medical Products Administration revealed late Friday that it’s granted conditional approval for Shanghai Green Valley Pharma’s oligomannate capsules to improve cognitive functions in patients with mild-to-moderate Alzheimer’s, finding an overnight oasis in a therapeutic desert. The decision, which followed a fast-track review, was based on a Phase III win that the company has yet to dissect in detail.
You may well not have heard of Green Valley — which was founded in 1997 to develop carbohydrate drugs and headquartered in Shanghai’s buzzing Zhangjiang Science City — but they have allied themselves with some familiar names for this Alzheimer’s program. IQVIA and Signant Health (formerly Bracket) were two of the CROs engaged in the Phase III program supporting the OK, and Eric Reiman — executive director of Banner Alzheimer’s Institute, which has been involved in key studies — is a scientific adviser alongside renowned researchers Jeffrey Cummings and Philip Scheltens.
Green Valley says the manuscript of that China trial is under review by New England Journal of Medicine, with a decision pending later this year. It also plans to initiate a second, global Phase III in early 2020, paving the way for submissions in the US, Europe and other Asian countries.
The consequences of a surprise win here can’t be overstated. Any new drug that can bend the course of the memory obliterating disease — after more than a decade of R&D drought in the field — is usually assigned a blockbuster peak sales estimate of $10 billion-plus. So this drug immediately becomes one of the most closely watched commodities in the pipeline. It’s also likely to inspire considerable skepticism.
As Chinese regulators stopped short of providing a full throated endorsement, Green Valley will have to submit follow-up data regarding pharmacology as well as long term safety and efficacy. But it plans to begin production as soon as November 7, CEO Songtao Lu told local reporters, and make the drug available nationwide by the end of the year.
“Reimbursement is expected from the government. The price point is not fully decided but it is expected to price at a premium of at least 2x acetylcholinesterase inhibitors given their limited efficacy,” a spokesperson told Endpoints News.
Reiman added that oligomannate — a seaweed-derived oligosaccharide known also as GV-971 — has the potential to diversify the portfolio of promising Alzheimer’s treatments.
Up until Biogen scrapped two big Phase III studies on aducanumab this March the amyloid beta theory remained the dominant, if fading, strategy to halt or at least slow down the neurodegenerative disease (and the big biotech revived some of those hopes recently in a controversial turnaround). But others have proposed that inflammation, specifically the type triggered by an imbalance of microbiota, plays a role in the ailment via the poorly understood bidirectional gut-brain axis.
Here’s how Meiyu Geng a professor at Shanghai Institute of Materia Medica and inventor of the drug, and her team described the mechanism of action in a Nature Cell Research paper about their preclinical work:
We have revealed that gut microbiota patterns and amino acid-derived metabolites are important for the infiltration of specific types of immune cells, which drives neuroinflammation during AD progression. GV-971 therapeutically harnesses the abnormal production of amino acids, infiltration of immune cells to the brain, and in turn neuroinflammation via remodelling the gut microbiota.
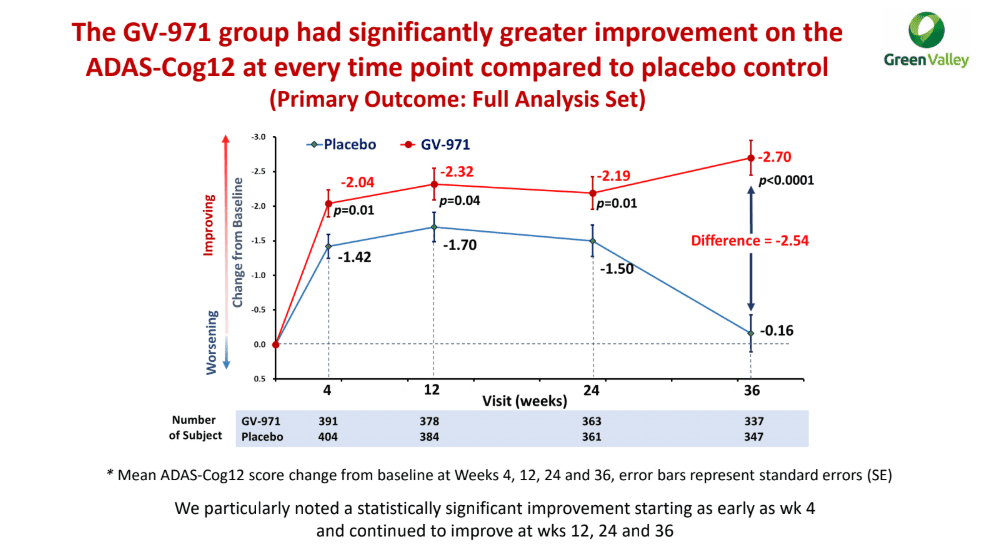
The theory apparently came through in a Phase III trial involving 788 Chinese patients, with the company touting a statistically significant improvement on the primary endpoint of ADAS-Cog12 (p<0.0001). Without computing the percentage change from baseline, Green Valley added that the mean difference the drug and placebo at week 36 was 2.54. None of the secondary endpoints — including CIBIC-plus, ADCS-ADL or NPI — were met.
“What is interesting about this is that Alzheimer’s is a progressive disease that unfortunately accelerates,” the spokesperson wrote in an email. “Realistically, the disease only progresses a little bit in 36 weeks for the less severe patients. But, it moves much faster for the later stage patients – creating more potential for separation between the active arm and control arm in a 36 week study. This is what was seen.”
Author
Amber Tong
ASSOCIATE EDITOR
Amber Tong
China
Regulatory
UPDATED: Alzheimer's stunner: Chinese biotech wins OK for a new drug on positive data — and they have some high-profile supporters
Did an obscure Chinese biotech just crack the Alzheimer’s enigma that’s dumbfounded a host of major league drug developers?
China’s National Medical Products Administration revealed late Friday that it’s granted conditional approval for Shanghai Green Valley Pharma’s oligomannate capsules to improve cognitive functions in patients with mild-to-moderate Alzheimer’s, finding an overnight oasis in a therapeutic desert. The decision, which followed a fast-track review, was based on a Phase III win that the company has yet to dissect in detail.
You may well not have heard of Green Valley — which was founded in 1997 to develop carbohydrate drugs and headquartered in Shanghai’s buzzing Zhangjiang Science City — but they have allied themselves with some familiar names for this Alzheimer’s program. IQVIA and Signant Health (formerly Bracket) were two of the CROs engaged in the Phase III program supporting the OK, and Eric Reiman — executive director of Banner Alzheimer’s Institute, which has been involved in key studies — is a scientific adviser alongside renowned researchers Jeffrey Cummings and Philip Scheltens.
Green Valley says the manuscript of that China trial is under review by New England Journal of Medicine, with a decision pending later this year. It also plans to initiate a second, global Phase III in early 2020, paving the way for submissions in the US, Europe and other Asian countries.
The consequences of a surprise win here can’t be overstated. Any new drug that can bend the course of the memory obliterating disease — after more than a decade of R&D drought in the field — is usually assigned a blockbuster peak sales estimate of $10 billion-plus. So this drug immediately becomes one of the most closely watched commodities in the pipeline. It’s also likely to inspire considerable skepticism.
“The phase 3 clinical trial of Oligomannate conducted in China showed a sustainable cognitive benefit. It was well tolerated,” Cummings, founding director of the Cleveland Clinic Lou Ruvo Center for Brain Health, said in a statement. “This is the first new therapy for Alzheimer’s disease approved in many years and we applaud this innovation.”

As Chinese regulators stopped short of providing a full throated endorsement, Green Valley will have to submit follow-up data regarding pharmacology as well as long term safety and efficacy. But it plans to begin production as soon as November 7, CEO Songtao Lu told local reporters, and make the drug available nationwide by the end of the year.
“Reimbursement is expected from the government. The price point is not fully decided but it is expected to price at a premium of at least 2x acetylcholinesterase inhibitors given their limited efficacy,” a spokesperson told Endpoints News.
Reiman added that oligomannate — a seaweed-derived oligosaccharide known also as GV-971 — has the potential to diversify the portfolio of promising Alzheimer’s treatments.
Up until Biogen scrapped two big Phase III studies on aducanumab this March the amyloid beta theory remained the dominant, if fading, strategy to halt or at least slow down the neurodegenerative disease (and the big biotech revived some of those hopes recently in a controversial turnaround). But others have proposed that inflammation, specifically the type triggered by an imbalance of microbiota, plays a role in the ailment via the poorly understood bidirectional gut-brain axis.
Here’s how Meiyu Geng a professor at Shanghai Institute of Materia Medica and inventor of the drug, and her team described the mechanism of action in a Nature Cell Research paper about their preclinical work:
We have revealed that gut microbiota patterns and amino acid-derived metabolites are important for the infiltration of specific types of immune cells, which drives neuroinflammation during AD progression. GV-971 therapeutically harnesses the abnormal production of amino acids, infiltration of immune cells to the brain, and in turn neuroinflammation via remodelling the gut microbiota.
The theory apparently came through in a Phase III trial involving 788 Chinese patients, with the company touting a statistically significant improvement on the primary endpoint of ADAS-Cog12 (p<0.0001). Without computing the percentage change from baseline, Green Valley added that the mean difference the drug and placebo at week 36 was 2.54. None of the secondary endpoints — including CIBIC-plus, ADCS-ADL or NPI — were met.
“What is interesting about this is that Alzheimer’s is a progressive disease that unfortunately accelerates,” the spokesperson wrote in an email. “Realistically, the disease only progresses a little bit in 36 weeks for the less severe patients. But, it moves much faster for the later stage patients – creating more potential for separation between the active arm and control arm in a 36 week study. This is what was seen.”
Author
Amber Tong
ASSOCIATE EDITOR






